Maltose PH

About Maltose PH
Maltose is a disaccharide consisting of two glucose molecules in a- 1,4 glucosidic bond and is therefore twice as efficient as glucose in supplying calories in an intravenous drips. Furthermore it is metabolised in the body with slight need for insulin, making it an ideal energy source for diabetics and patients with temporary post-operative diabetes. It has little influence on blood-sugar level and favourably influences the metabolism of lipids and proteins. Maltose PH is an odourless, white crystalline powder. In addition to its use for nutritional intravenous drip, it can be also used as an excipient/stabilizer for incorporation into various pharmaceutical drug or biologic products.
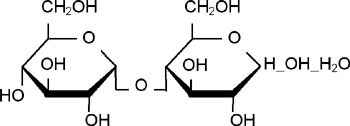
[ Chemical Structure of Maltose ]
[ Generic Name ] Maltose monohydrate
[ Chemical Name ] 4-O- a-D-Glucopyranosyl-D-Glucopyranose monohydrate
[ Molecular Weight ] C12 H22O11H2O: 360.31
Maltose is listed in the Thirteen Edition of the Japanese Pharmacopoeia. Approval has been granted by the Japanese Minister of Health and Welfare for the use of a 10 % solution of Maltose PH as an intravenous drip for an energy (sugar) source during or after operations and for diabetes mellitus patients.
In the United States, Maltose PH was registered for a DMF (Drug Master File) in October 2001.
Note: All pictures & informations are for reference only. The actual goods in kind prevail.
 After send online enquiry, we will reply you as soon as possible, if not get any response on time please contact us by Tel or Email.
After send online enquiry, we will reply you as soon as possible, if not get any response on time please contact us by Tel or Email.
Welcome to Bolise Co., Ltd.
1. E-mail: [email protected]
2. Tel: +86 592 5365887
Working Time: 8:30--18:00, Monday--Friday
For more information of product, please send the email to [email protected]

- 1. Send Message to US ->
- 2. Confirm Product Information ->
- 3. Order & Payment->
- 4. Packaging&Shipping->
Our products have been certificated with the ISO certificate, free sample is available.
CGMP Manufacturer to ensure the quality stable and reliable.
All of our ingredients meet strict quality and food safety standards, and surpass industry standards for purity and cleanliness.
We are committed to meeting customers' unique needs by providing comprehensive and professional service, enjoying a good reputation among our business partners and customers for high-quality products, excellent after-sales services, competitive prices, and prompt shipping.
Highly welcome friends from all over the world to contact us. We will reply to you as soon as possible, after sending an online inquiry. And please feel free to call us if any urgent inquiry or without getting response from us in time.

 Asia Bio-Pharmaceutical Research Institute
Asia Bio-Pharmaceutical Research Institute Bolise Co., Ltd.
Bolise Co., Ltd.